We invite all health care professionals (physicians, allied health professionals, individuals outside the health care field) with an interest in pediatric bioethics to join the American Academy of Pediatrics Section of Bioethics. If you are not eligible to be a member of the AAP, you can still join our section as a Section Affiliate Member for a small membership fee.
We welcome all to be part of a vibrant pediatric ethics community, receive our newsletter, and propose education programs for a national meeting.
A journal devoted to pediatric bioethics
space
In Press
Ethically difficult situations often occur in the treatment of sick children and can be associated with conflicting opinions among members of the health care team and parents about what represents the patient’s best interests. This study aimed to identify if differences exist between the perspectives of parents, nurses, chaplains, and physicians.
Legal and social precedent support broad parental discretion in the absence of immediate and severe endangerment of the minor’s welfare. However, in many cases of maltreatment, suspected or substantiated, parents retain PDM rights. The possibility of a guardian who is both surrogate and cause of harm threatens the conventional decision-making model; this paper offers an analysis of the primary challenges and shows that many of the ethical questions arise as the decision-making process unfolds and information is gathered.
Recent Articles
Ethically difficult situations often occur in the treatment of sick children and can be associated with conflicting opinions among members of the health care team and parents about what represents the patient’s best interests. This study aimed to identify if differences exist between the perspectives of parents, nurses, chaplains, and physicians.
Legal and social precedent support broad parental discretion in the absence of immediate and severe endangerment of the minor’s welfare. However, in many cases of maltreatment, suspected or substantiated, parents retain PDM rights. The possibility of a guardian who is both surrogate and cause of harm threatens the conventional decision-making model; this paper offers an analysis of the primary challenges and shows that many of the ethical questions arise as the decision-making process unfolds and information is gathered.
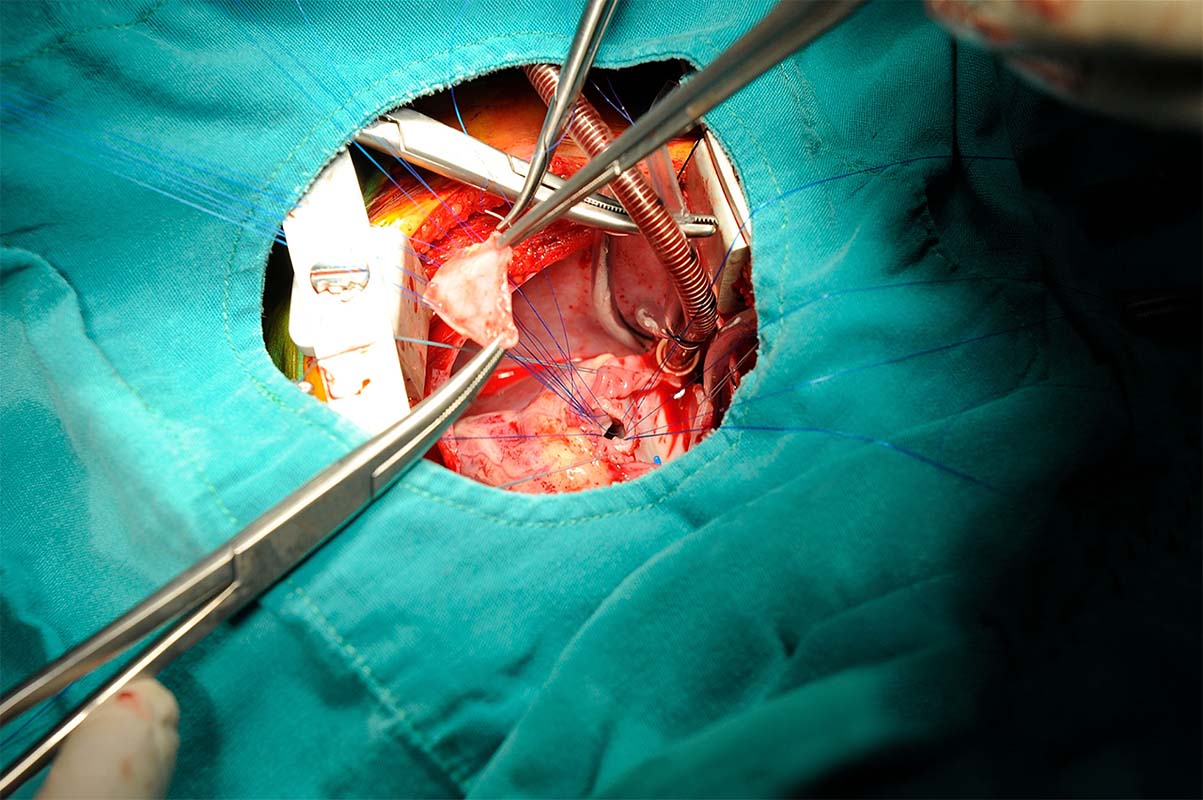
Death and Scandal in Pediatric Cardiac Surgery Programs
Over the past few years, several high-profile scandals have befallen well-known U.S. pediatric cardiac surgery programs. The aftermath paints a picture of persistent systemic institutional problems that motivated staff to become whistleblowers. The industry experts point to the lack of regionalization as the problem. Is the cause of such problems the lack of regionalized pediatric cardiac surgery centers? We analyze recent U.S. scandals to contrast and compare them with international scandals and approaches to pediatric cardiac surgery.
Life-Prolonging Therapies in a Case of Anencephaly: A Mother’s Wish
Medical futility is often a point of debate in situations of hoping for an unlikely outcome, or a condition with a treatment that necessitates high levels of pain or discomfort. Anencephaly is generally accepted as incompatible with prolonged life, with few infants surviving into the neonatal period. Parents are typically counseled accordingly, resulting in pregnancy termination or comfort care after delivery. This case report examines the ethical considerations involved in the decision to continue life-prolonging treatment of an 8-month-old infant with anencephaly.
Refugee Children: The Tiny Victims of Huge Human Catastrophes
Offering sanctuary to those fleeing war is a pressing human rights issues in the world today. Globally, there are currently 25.4 million people forced to leave their home to seek safety. Half of these people are children. Despite these staggering statistics, governments seem to have ignored their duty of care to children.
In a field dominated by analytic thinking, taking a moment to consider the aesthetic and emotional contours of a subject can shed new light on it. The Proem serves as an introductory thought that relates to an idea explored in one or more of the articles in the issue.
Psychological and Medical Evaluations: Asylum Seekers, Ethics, and the Law
This article highlights the importance of psychological and medical evaluations for asylum seekers in the United States, and identifies physicians and other healthcare professionals as uniquely situated for this work. This paper outlines the benefits and drawbacks to such evaluations and addresses their utility in immigration law, ultimately calling for increased clinician involvement in pro bono evaluations.
Conceptual Considerations for Trainees in Asylum Medicine
In recent years, the unique role of medical professionals in the asylum adjudication process has been thrown into sharp relief as asylum applications surge, with over one million pending cases backlogged in the U.S. asylum system as of August 2019. Medical evaluations dramatically increase the likelihood of an individual obtaining asylum. The author examines the role medical trainees play in this process.
Ethiopian physicians, nurses, and midwives routinely encounter cultural challenges created by language barriers, an urban vs rural divide, and differences in education that impact the patient-provider relationship. Despite limitations in personnel and resources, these clinicians have devised approaches to overcome these barriers to best serve their patients.
For Clinical Ethicists
space
HEC Analytics is a new project initiated at the 2022 UnConference, Propelling Clinical Ethics Forward, hosted by Wellstar Health System in Atlanta, GA. The purpose of the project is to share consult metrics across institutions and health systems for quality improvement, benchmarking, and development of the profession. Pediatric Ethicscope is hosting the project site, accessible at right.
Archived Announcements

Dear readers, each year the American Academy of Pediatrics Section on Bioethics presents the Academy’s highest honor in bioethics. It is our pleasure to share that Pediatric Ethicscope founder and editor emeritus, Tomas Silber MD, MAAS, (at left) has been awarded that honor, the William G. Bartholome Award for Ethical Excellence. You can read Dr. Silber’s recent Leikin Lecture, “On Suffering,” here.